Expansion announced after groundbreaking highlights why life sciences are diverging from stalled EV projects
Содержание:
Genentech on Jan. 20 more than doubled its planned investment in a biomanufacturing campus in Holly Springs, N.C., expanding a project that was first announced in May 2025 and broke ground last summer.
The move lifts the facility to roughly $2 billion and materially increases the scope of construction already underway, offering a real-time test of whether biomanufacturing projects can continue to scale smoothly amid tightening infrastructure and delivery constraints.
“This expansion reflects our long-term commitment to the United States and communities like Holly Springs,” Genentech CEO Ashley Magargee said in a statement. “This additional investment will create more high-quality jobs, strengthen local partnerships, and ensure a resilient supply of medicines for years to come.”
2026 Market Report
Manufacturing Momentum | Newmark Industrial Research
The expansion isn’t a new project announcement; it reflects an increase in capital for work already underway. Genentech has not yet disclosed revised square footage, building counts or construction phasing tied to the increase, but the added capital places the campus among the larger active biomanufacturing builds now underway in the U.S., reshaping delivery considerations already in motion.
An analysis released Jan. 26 by Newmark Research found that pharmaceutical and biomanufacturing projects posted the strongest investment growth among major advanced manufacturing sectors in 2025, driven by long-horizon demand, supply-chain resilience goals and regulatory barriers that discourage rapid relocation once facilities are operational.
For builders, that durability comes with complexity. Biomanufacturing facilities rank among the most infrastructure-intensive industrial projects delivered today, requiring validated cleanrooms, redundant power systems, precision HVAC and sophisticated water and wastewater treatment. Expanding scope after groundbreaking typically reshapes execution risk more than headline economics.
Power: Redundancy Becomes the Binding Constraint
On advanced manufacturing projects, power quality and redundancy are becoming more important than raw capacity, as the data center boom has demonstrated. Semiconductor fabs in Arizona and Texas have required dedicated substations and multi-year coordination with utilities, with transformer procurement becoming schedule-critical.
While biomanufacturing loads are generally lower, facilities of this scale often require dual-feed service, backup generation and uninterrupted power for process-critical systems.
To accommodate future phases, owners frequently oversize central utility plants early in delivery, protecting long-term capacity but extending early design timelines and increasing upfront capital exposure. Whether Genentech’s expansion requires off-site utility upgrades or relies primarily on on-site redundancy has not been disclosed.
Water and Wastewater: A Quieter Pressure Point
In biomanufacturing, wastewater treatment often poses greater execution risk than water supply. Pharmaceutical production can generate complex effluent streams that exceed municipal discharge limits without pretreatment, pushing utility coordination and permitting into the critical path even when building approvals are already in hand.
Comparable life-sciences projects in North Carolina and other established hubs have required on-site pretreatment systems and prolonged engagement with local utilities, in some cases extending schedules as discharge characteristics and capacity thresholds are negotiated. Those challenges tend to intensify as projects scale, because wastewater systems are typically designed for peak future capacity rather than initial operations, leading to early decisions that impact site layout, underground work, and mechanical sequencing.
2026 Market Report
Watering the New Economy | Global Water Intelligence, Xylem
That dynamic is not unique to pharmaceuticals. A January analysis by Global Water Intelligence and Xylem examining how infrastructure-intensive industries are intersecting with municipal water systems found that the binding constraint is increasingly system adaptability rather than total supply.
“The biggest opportunity to increase water supply without raising freshwater withdrawals is wastewater reuse,” the report states, adding that major industrial operators are increasingly willing to invest in reuse projects with municipal utilities to secure supply and improve overall system resilience.
It is not yet clear whether Genentech’s expanded scope will require amended discharge permits or additional pretreatment capacity, but projects of similar scale have shown how wastewater considerations can become schedule-defining issues as construction advances.
RELATED
Construction’s Labor ‘Relief’ Masks Structural Risk as Demand Cools
Labor, Permitting and Sequencing Risk
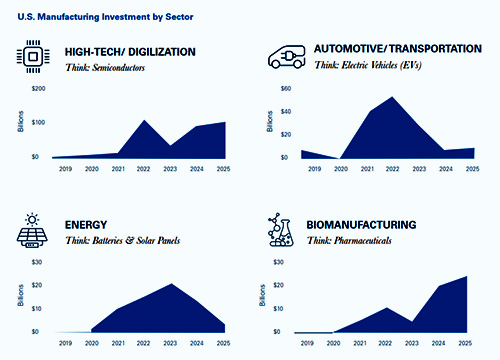
A Newmark Research analysis shows U.S. manufacturing investment shifting by sector since 2019, with biomanufacturing accelerating in recent years while electric vehicle investment has cooled. The data underscores why life sciences projects continue expanding even as other advanced manufacturing sectors recalibrate.
Infographic courtesy of Newmark Industrial Research
While national manufacturing employment contracted in 2025, markets hosting large manufacturing builds continued adding construction and supplier jobs. Newmark’s analysis shows ecosystem employment growth outpacing direct plant staffing in markets with major projects, reflecting sustained demand for skilled trades tied to facility buildout.
To manage labor constraints, contractors on recent megaprojects have staggered cleanroom installation, broken work into smaller packages and extended commissioning phases to smooth workforce demand. Those strategies protect quality but can stretch schedules, a key consideration when scope expands mid-delivery.
Expansions that materially increase throughput often trigger permit amendments even when zoning and land use remain unchanged. Owners increasingly pursue parallel early-works packages—site work, utilities and foundations—while detailed process design continues. That approach preserves momentum but adds coordination complexity.
Genentech has not yet indicated whether the Holly Springs expansion will proceed as a single integrated build or multiple phases, a decision that will shape contractor procurement, risk allocation and flexibility as process requirements evolve.
RELATED
2025 ENR West Best Projects: Genentech, Excellence in Safety, Interior/Tenant Improvement
A Bellwether for Resilient Reshoring
The Holly Springs expansion highlights how biomanufacturing is diverging from other reshoring sectors. While some manufacturing projects announced earlier in the decade have been delayed or downsized due to financing and market pressures, life-sciences facilities continue to move forward—and, in this case, expand after construction has begun.
For readers, the main question is not whether Genentech will build, but how effectively projects of this class can absorb scope growth as constraints on power, water, labor, and permitting become tighter.
As construction details emerge, the project will serve as a bellwether for whether biomanufacturing’s momentum translates into predictable, buildable work—or simply shifts where execution risk concentrates next.